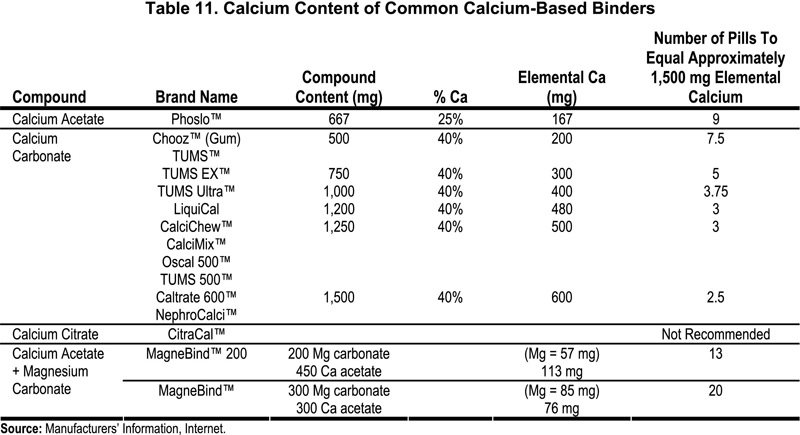
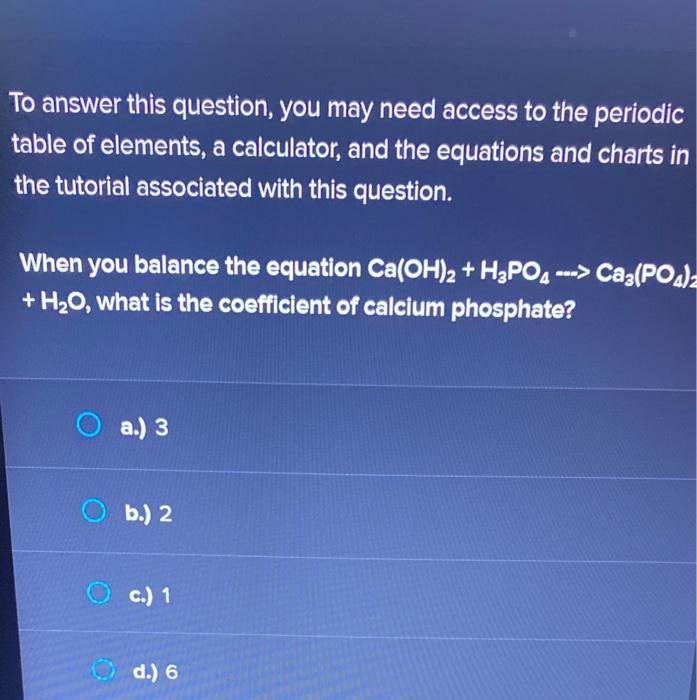
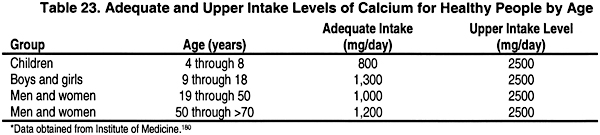
SOLVED: The desired product is calcium phosphate. How is the calcium phosphate collected after the reaction is completed? A. Filtration B. Evaporation C. Distillation D. Decantation A 1.520 g sample containing a

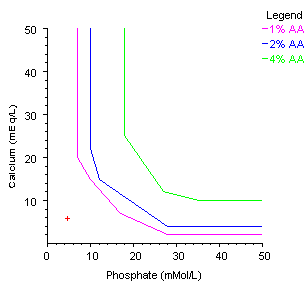
Calcium and Phosphate Solubility Curve Equation for Determining Precipitation Limits in Compounding Parenteral Nutrition - Collin Anderson, Larry Eggert, Kristie Fitzgerald, Daniel Jackson, Fred Farr, 2022